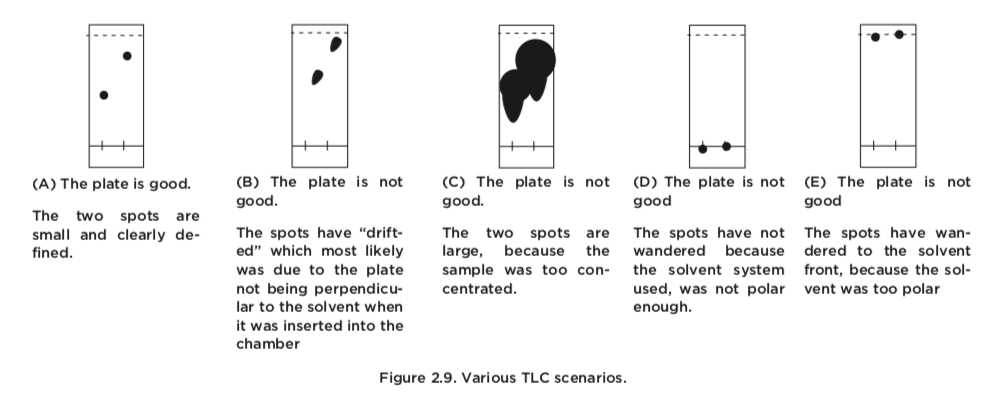
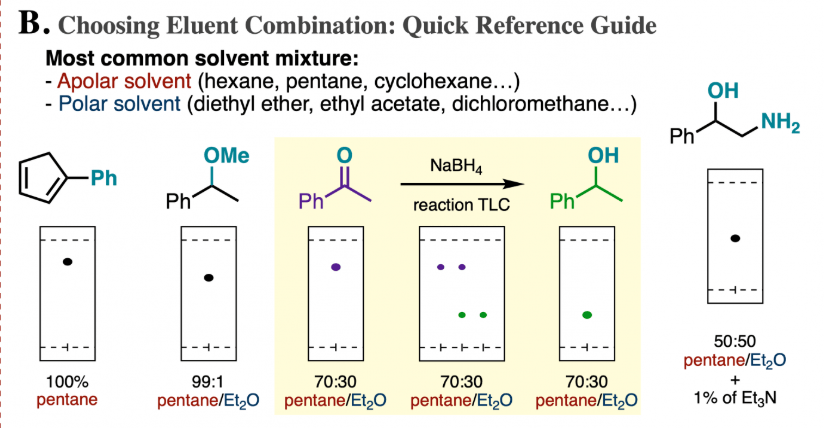
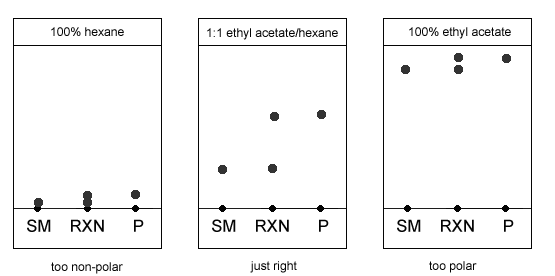
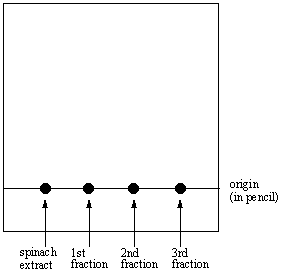
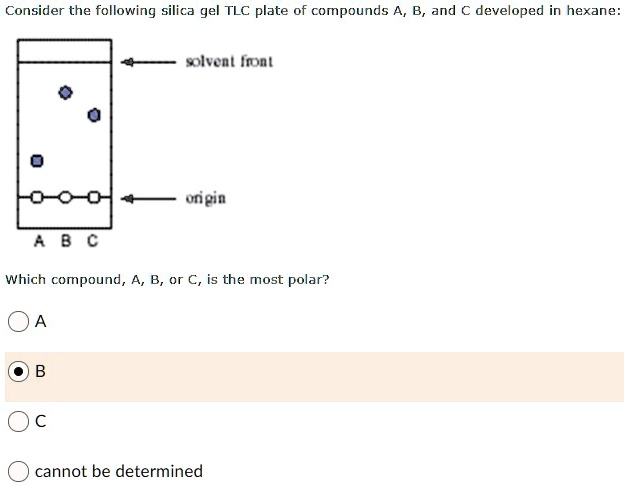
The TLC plate below was developed in an 80:20 hexane: acetone solvent system. (Image) a) Is a 50:50 hexane: acetone solvent system more or less polar than 80:20 hexane: acetone? b) What

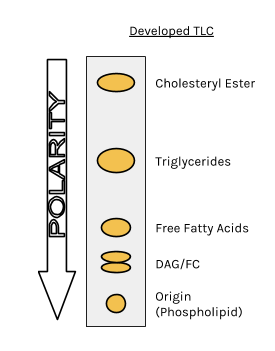
Do more polar molecules tend to have larger or smaller Rf values by TLC when compared to less polar molecules? | Homework.Study.com

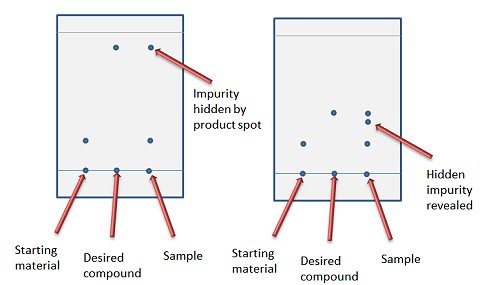
TLC and solvent polarity!! What does it mean that it's 'too non polar' and close to the origin? : r/Mcat

organic chemistry - How does the polarity of the eluent and sample affect the Rf value in thin layer chromatography? - Chemistry Stack Exchange
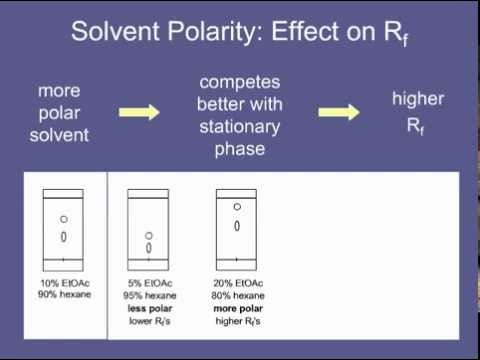
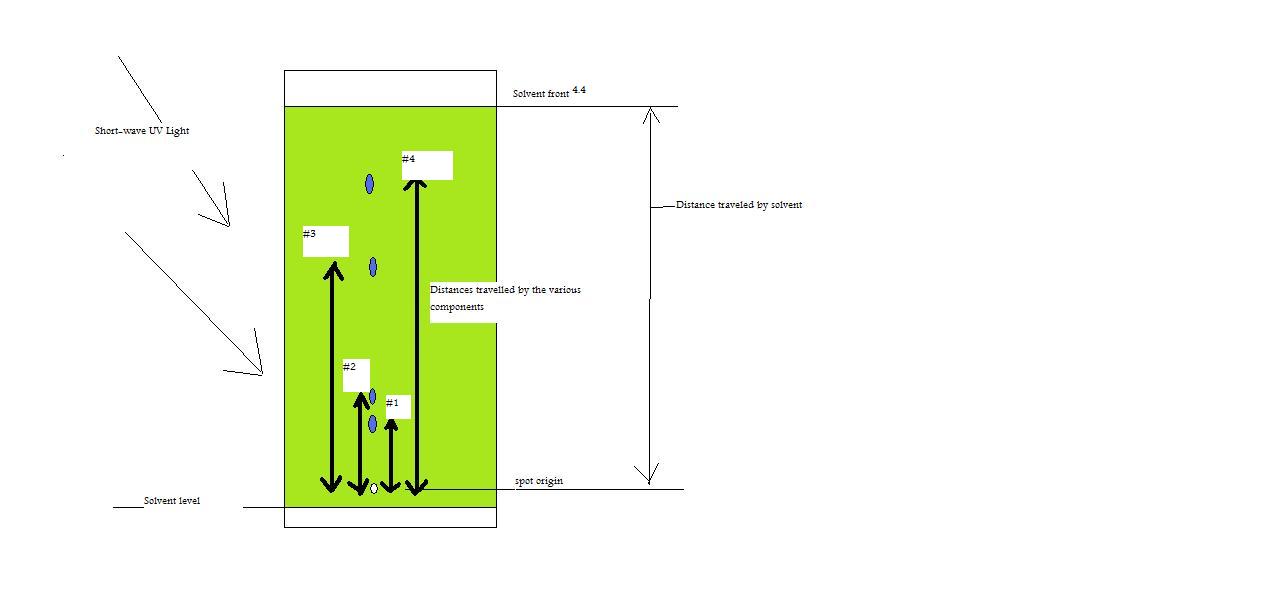
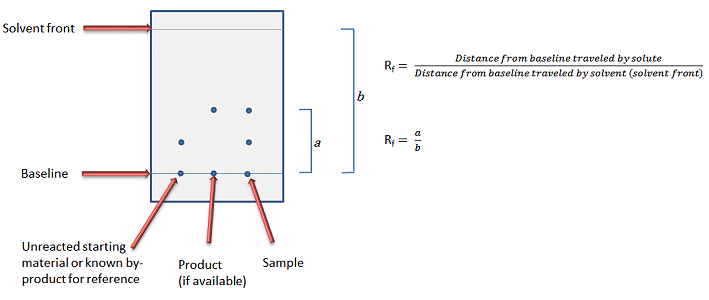
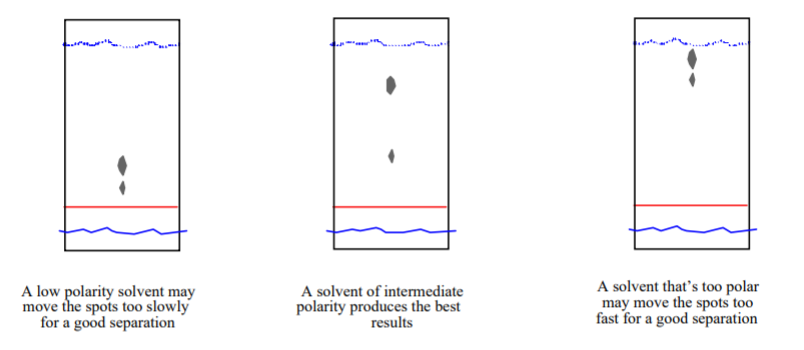

/chapter4/pages3and4/page3and4_files/rfvalue.png)
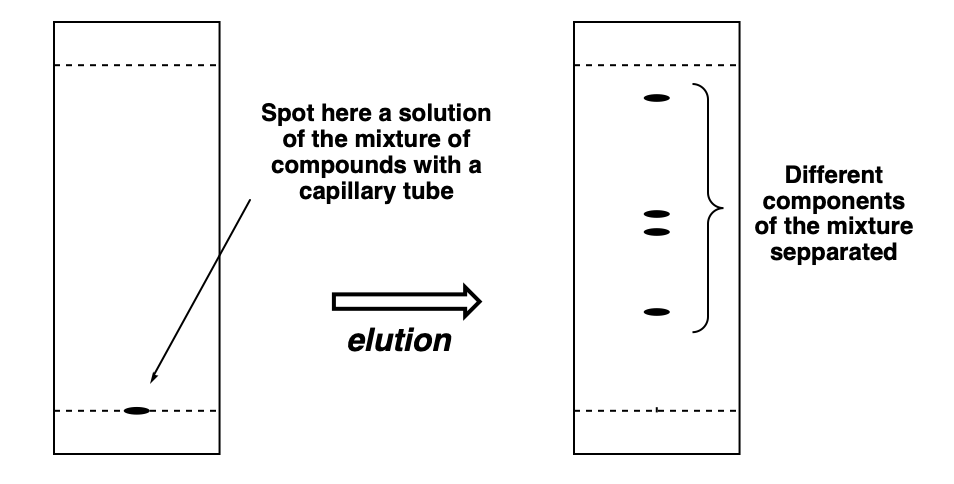
/chapter4/pages3and4/page3and4_files/tlcsetup.png)