SOLVED: Which formula/name pair is incorrect? FeSO4 - iron(II) sulfate FeSO3 - iron(II) sulfite Fe2(SO3)3 - iron(III) sulfite FeS - iron(II) sulfide Fe2(SO4)3 - iron(III) sulfide Moving to the next question prevents
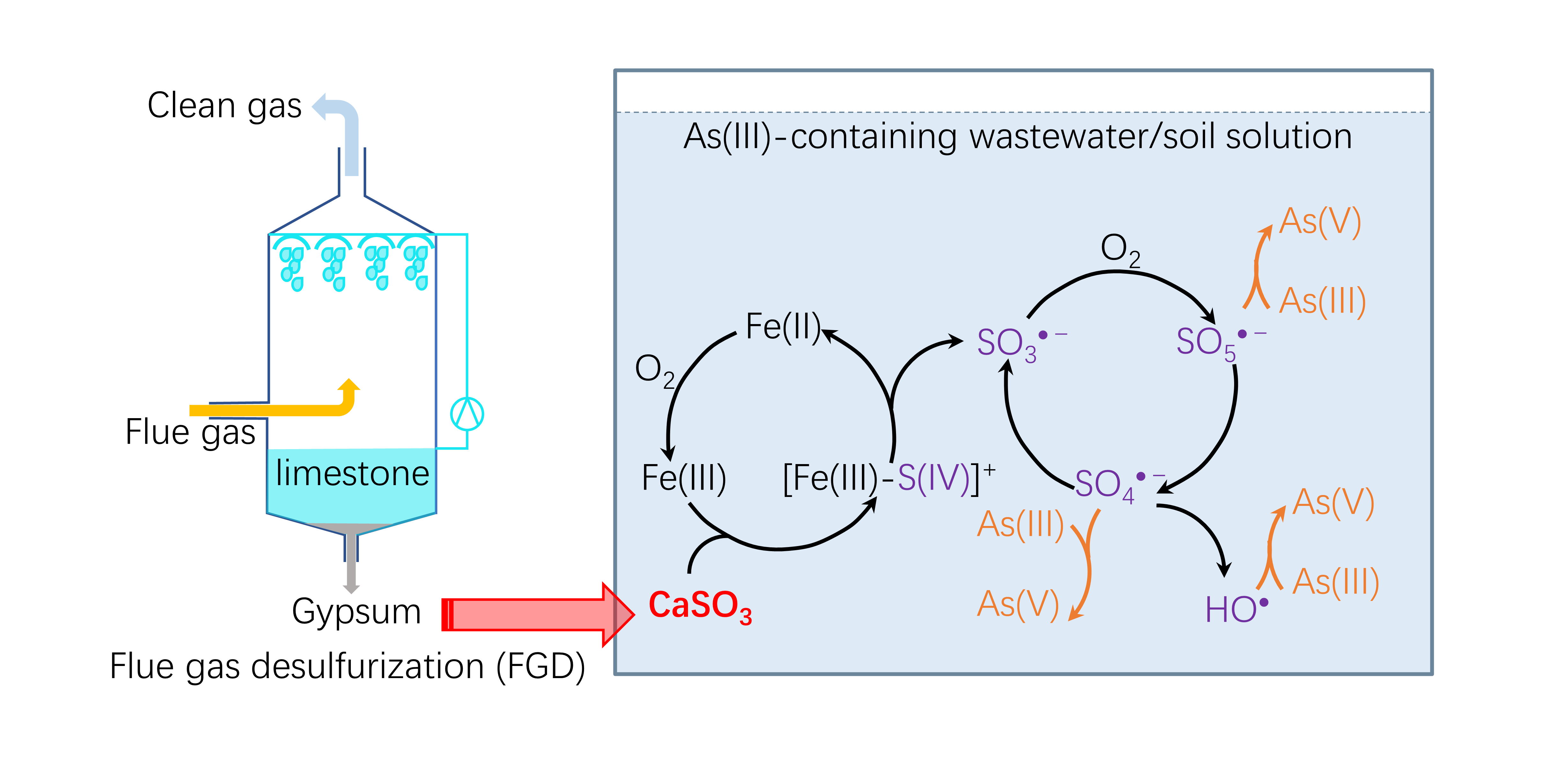
Molecules | Free Full-Text | Calcium Sulfite Solids Activated by Iron for Enhancing As(III) Oxidation in Water

Compounds vs. Elements Compound Table Salt : Soluble crystals, stable, edible Elements (Components) Sodium – shiny metal, reactive, poisonous Chlorine. - ppt download

The Early Phase of the Iron(III)−Sulfite Ion Reaction. Formation of a Novel Iron(III)−Sulfito Complex | Inorganic Chemistry